Company targets pivotal trials of brain-computer interface platform that allows control of digital devices using thought alone.
Neurotech company Synchron today announced it has secured $200 million in Series D financing to accelerate the commercialization of its brain-computer interface (BCI) platform, bringing its total funding raised to $345 million. In addition to supporting pivotal clinical trials, the company said the funding will be used to develop next-generation interfaces that aim to expand the scope of brain-computer communication.
Synchron develops minimally invasive BCIs, initially intended to restore communication and mobility for people with paralysis. The company’s endovascular approach, first tested in humans in 2019, sets itself apart from surgical implants that require removing sections of the skull. Its platform, designed to be placed within the brain’s blood vessels via catheter rather than through open-brain surgery, detects neural activity and converts it into digital signals. These signals are transmitted wirelessly to allow hands-free control of digital devices.
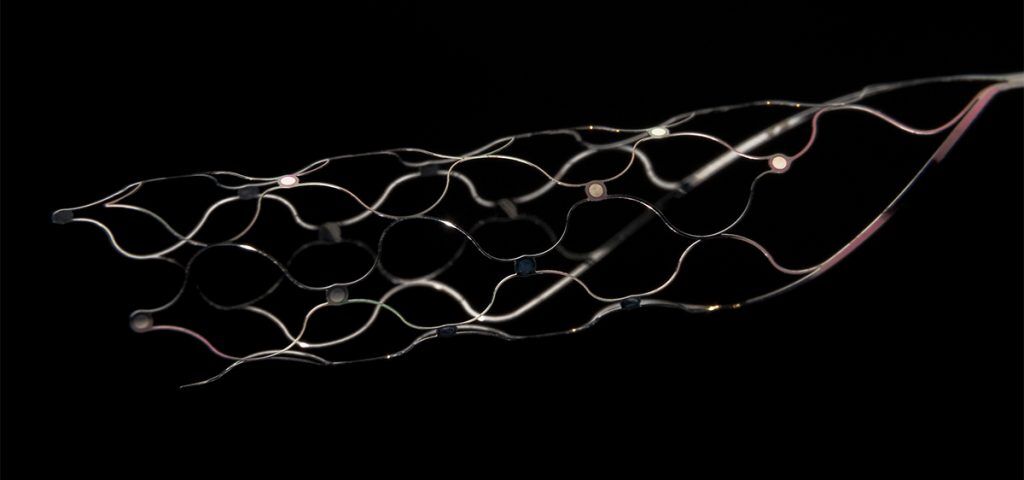
“We’ve built the first non-surgical brain-computer interface designed for everyday life for people with paralysis,” said Synchron founder Tom Oxley. “This funding brings us closer to commercializing the Stentrode BCI platform, while accelerating development of a major breakthrough in the field – a next-generation, transcatheter high-channel whole-brain interface.”
In terms of the company’s future direction, Oxley told us that Synchron’s technology could one day be used to help combat aging and age-related diseases.
“While our immediate focus is restoring digital agency to people with paralysis, the underlying platform has the potential to extend far beyond motor control,” he said. “We see a future where endovascular BCIs could also help with neurodegenerative diseases, for example cognitive and functional decline associated with aging, bringing brain health into the era of proactive, precision care.”
To date, Synchron’s BCIs have been implanted in ten patients through clinical trials in the United States and Australia. The system was the first of its kind to be integrated with Apple’s Brain-Computer Interface Human Interface Device (BCI-HID) protocol, enabling thought-driven control of iOS devices. Earlier this year, a patient with ALS used Synchron’s implant to operate an iPad solely by thought, performing actions such as navigating the home screen, launching apps and composing messages without touch, voice, or eye tracking.
The funding will support the company’s next phase of development, including pivotal clinical trials and preparations for the commercial launch of its BCI system. Synchron said it also plans to expand its workforce of engineers, neuroscientists and AI specialists to advance its technology pipeline. The company’s Cognitive AI division in New York is developing models that interpret neural activity in real time, while its newly established San Diego engineering hub is focused on creating higher-channel brain interfaces capable of capturing more comprehensive brain data.

The Series D financing is also intended to accelerate both the path toward commercialization of the current Stentrode system and the development of
The round, led by Double Point Ventures with participation from new and existing investors including ARCH Ventures, Khosla Ventures, Bezos Expeditions, NTI, METIS, the Australian National Reconstruction Fund, T.Rx Capital, Qatar Investment Authority, K5 Global, Protocol Labs and IQT,
“Synchron is building the first truly scalable, minimally invasive brain-computer interface that can be deployed in everyday healthcare,” said Double Point co-founder Campbell Murray. “Its fusion of neurovascular access, breakthrough device engineering, and adaptive AI marks a fundamental step toward restoring digital agency to people with paralysis.”


