Founder and CEO Kevin Slawin MD recommends new, more precise criteria for defining ‘senolytic’ therapeutics.
In 2017, Ponce Therapeutics, Inc was founded in Houston, TX focused on developing gerotherapeutics to improve healthspan and increase lifespan. In October 2024, the company’s dramatic discoveries and innovative technologies in the fields of geroscience and longevity medicine led to the renaming of Ponce Therapeutics, Inc to SENOTHERAPEUTIX, Inc and its reorganization into a flexible ‘hub and spoke’ corporate structure.
Both Juvenescence and Cambrian Biopharma, top companies in the longevity space, are also organized in ways that resemble the hub-and-spoke model, though each has its own unique interpretation of it.
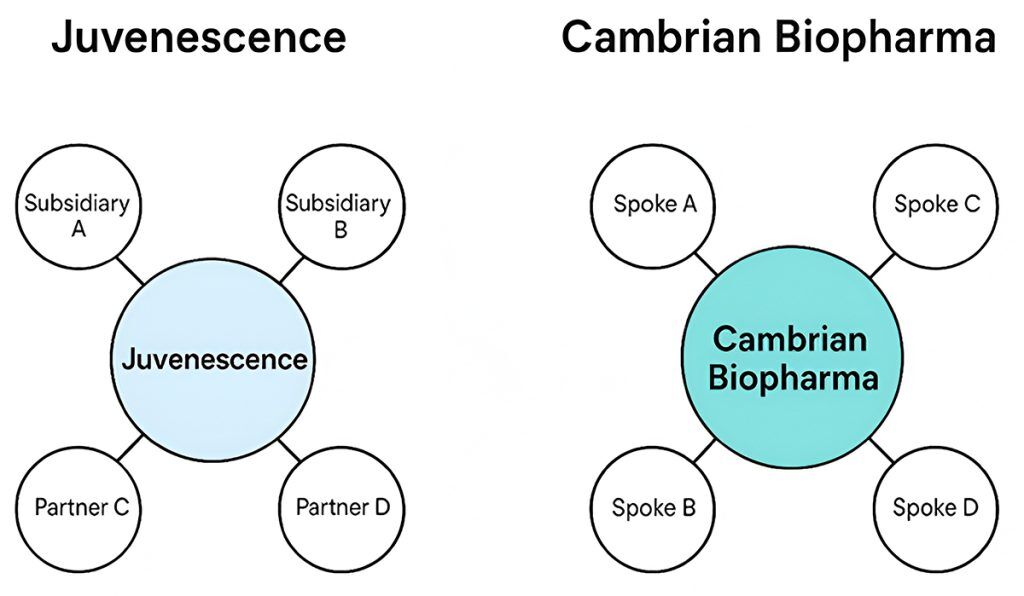
Cambrian explicitly describes its structure as a hub-and-spoke model, which they refer to as a DisCo (Distributed Company) model. Here’s how it works:
- Cambrian acts as the central hub, providing core resources such as drug discovery expertise, operational support, and funding.
- Spoke companies are individual biotech ventures focused on specific therapeutic targets or aging-related diseases.
- Cambrian is often the majority shareholder in these companies, such as Sensei Bio, and helps guide their development while allowing them operational independence.
- This model allows Cambrian to rapidly scale, de-risk early-stage biotech ventures, and leverage shared resources across its portfolio.
Juvenescence also operates in a way that aligns with the hub-and-spoke concept:
- It invests in and partners with multiple biotech companies, research institutions, and scientists globally.
- Juvenescence provides centralized leadership, funding, and strategic direction, while its partners and subsidiaries focus on specific therapeutic areas or technologies.
- The company blends biotech innovation with AI, and its structure allows it to diversify risk and accelerate development across multiple aging-related therapies.
Companies organized in this way use a centralized parent entity to manage and support a portfolio of specialized subsidiaries or affiliates, which is the hallmark of the hub-and-spoke model. This structure is increasingly popular in biotech because it enables focused innovation, efficient resource allocation, and flexible fundraising, including:
- Risk Diversification
a. Multiple spokes (subsidiaries or ventures) allow the parent company to spread risk across different therapeutic areas or technologies.
b. If one project fails, others may still succeed, protecting overall company value. - Capital efficiency
a. Centralized resources (the hub) such as legal, HR, finance, and R&D infrastructure reduce duplication and lower costs.
b. Spokes can operate leaner and focus on science and product development. - Accelerated innovation
a. Each spoke can pursue high-risk, high-reward ideas independently.
b. The hub provides strategic guidance and funding, enabling faster iteration and development. - Flexible exit strategies
a. Spoke companies can be spun out, sold, or IPOed individually.
b. This creates multiple potential liquidity events for investors and the parent company. - Strategic focus
a. The hub maintains a long-term vision (for example, combating aging), while spokes focus on specific scientific or commercial goals.
b. This balance allows for both visionary leadership and tactical execution.
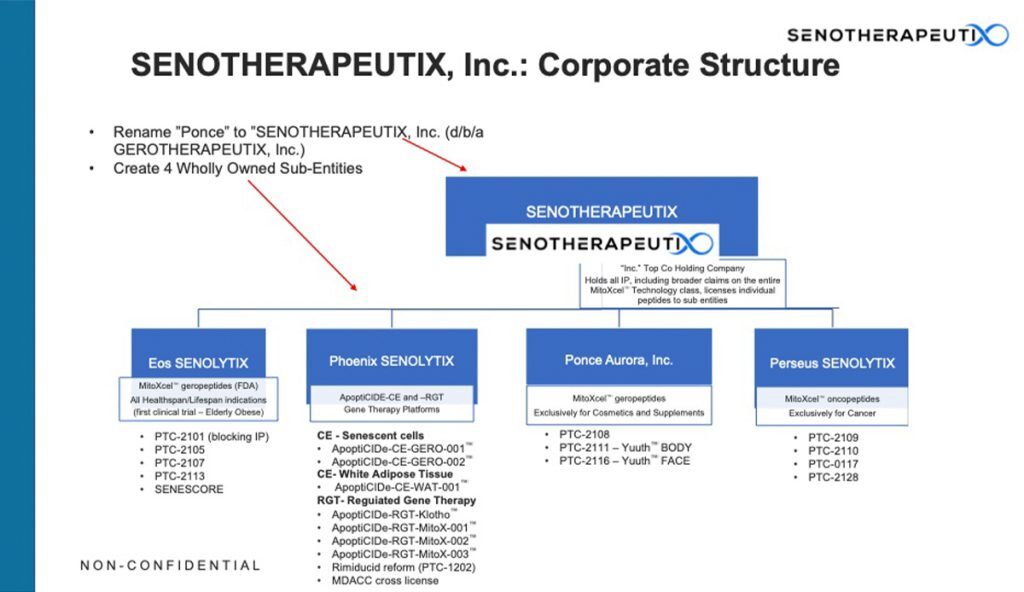
The renamed corporate entity, originally Ponce Therapeutics, Inc and now SENOTHERAPEUTIX, Inc, established “d/b/a GEROTHERAPEUTIX, Inc”, was established as the ‘hub’ holding and operating company, which also files and holds all Intellectual Property, including that on MitoXcel Technology, and sublicenses individual candidate therapeutics to the appropriate sub entities.
Four new wholly owned subsidiary corporate entities of GEROTHERAPEUTIX, Inc or ‘spokes’ were established:
- Eos SENOLYTIX, Inc – a longevity company developing novel MitoXcel geropeptides to improve healthspan and increase lifespan. It licenses MitoXcel geropeptides exclusively for all FDA-regulated healthspan and lifespan indications , and its first clinical trial will be in the elderly obese.
a. “Composition of Matter” for clinical candidates including PTC-2101, PTC-2105, PTC-2107, PTC-2113 and SENESCORE. - Phoenix SENOLYTIX, Inc – developing novel, controllable gene therapies using our proprietary ApoptiCIDe purposeful cell elimination technology to improve healthspan and increase lifespan. It licenses ApoptiCIDE-CE and –RGT gene therapy platforms exclusively, including PTC-1202 (SQ/IM Rimiducid).
a. Party to the MDACC Cross License. - Ponce Aurora, Inc – a longevity company developing novel cosmetics that help to reverse the signs of aging. Our lead product, Yuuth is powered by MitoXcel Technology, the first information- and AI-driven geropeptides aimed at an aging-specific target. Yuuth is engineered to eliminate or neutralize the cells that cause inflammaging in the skin but also throughout the body, as well as re-energize and rejuvenate aging cells. Yuuth, including all of its components, is FDA-regulated as a cosmetic and certified as never having been tested at any stage of product development on animals. Ponce Auroroa licenses MitoXcel geropeptides exclusively for cosmetic and supplement products.
a. “Composition of Matter” for PTC-2108, PTC-2111 (Yuuth BODY) and PTC-2116 (Yuuth FACE). - Perseus SENOLYTIX, Inc – an oncology company developing novel MitoXcel™ oncopeptides to target cancer via this same novel mechanism as a potentially safe and effective new class of cancer therapeutics. It licenses MitoXcel oncopeptides exclusively for the treatment of cancer.
a. “Composition of Matter” for PTC-2109, PTC-2110, PTC-0117 and PTC-2128.
All four sub entities of SENOTHERAPEUTIX, Inc/GEROTHERAPEUTIX, Inc already hold valuable assets and expect to raise capital independently in 2026.
SENOTHERAPEUTIX, Inc/GEROTHERAPEUTIX, Inc is physician-led by Founder and CEO Kevin Slawin, MD, an experienced leader with over 30 years of biotech experience, including as founder of Bellicum Pharmaceuticals in 2004, the first of the original three CAR-T cell companies, that he took public in an IPO on NASDAQ in 2014, at a $500M market cap. The Company operates from its state of the art R&D facility, that includes an in house mouse vivarium and a full range of advanced research equipment tailored for longevity experiments, including DEXA scanner, Rotarod, Y-maze, motorized treadmill, and others, at K2Bio in Houston, TX.
Focus on MitoXcel Technology
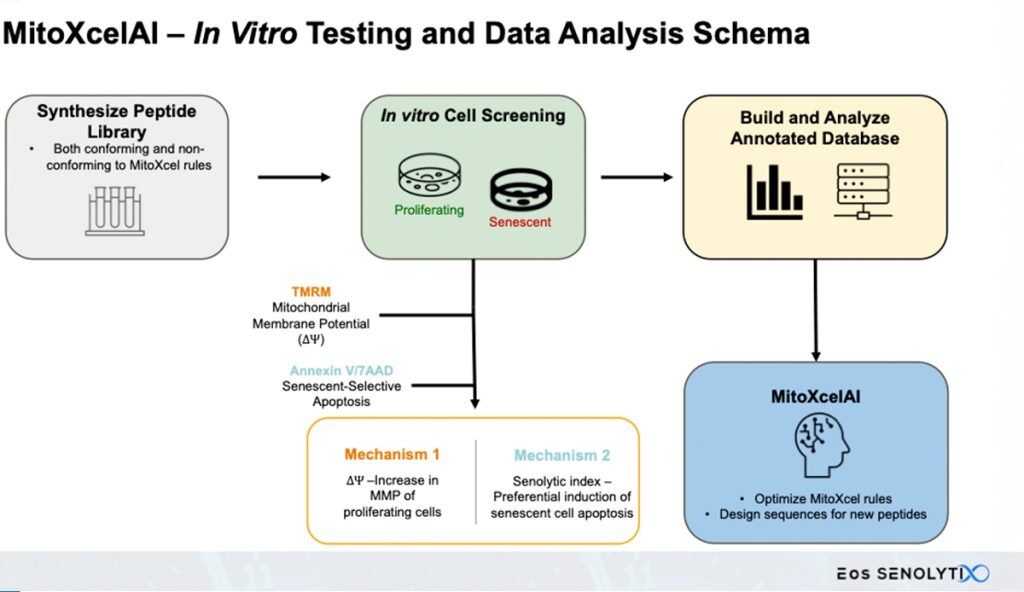
While traditional pharmaceuticals act by binding to specific receptors or regulatory molecules to alter biological functions, MitoXcel Technology introduces a revolutionary new class of peptide therapeutics whose effects are determined by the informational content of their amino acid sequences – much like DNA conveys genetic instructions through its nucleotide sequence. Instead of relying on a single, defined chemical entity, MitoXcel Technology is a powerful family of peptides – each precisely engineered to meet a unique set of design rules based on their amino acid sequences. Only those that match the MitoXcel blueprint qualify, ensuring consistent performance at the molecular level. Thus, MitoXcel peptides are the first ever information-based gerotherapeutics, specifically engineered to target the age-related decline in Mitochondrial Membrane Potential (Δψm) observed across all living organisms. By addressing this decline, MitoXcel reverses aging-related changes through two distinct mitochondrial-mediated mechanisms of action.
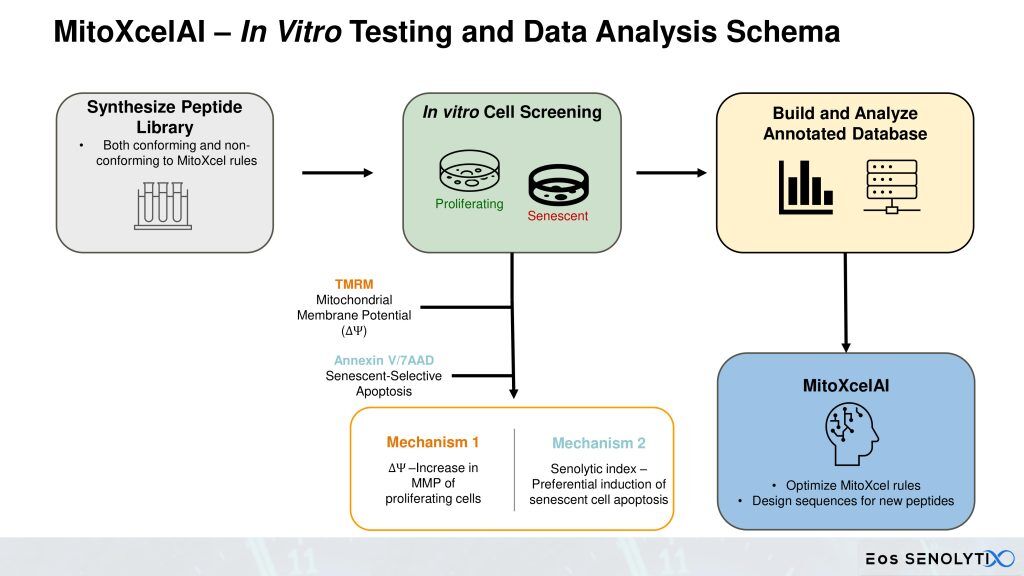
SENOTHERAPEUTIX, Inc is actively expanding a comprehensive database that links specific peptide sequences to preclinical outcomes related to aging reversal. Through an iterative learning process, MitoXcelAI – the platform’s proprietary artificial intelligence engine – analyzes this growing dataset to train and refine predictive algorithms. This continuous feedback loop enhances the rules that define MitoXcel Technology, ensuring its evolution as a best-in-class, information-engineered solution for combating age-related decline.
Eos SENOLYTIX’s PTC-2015, PTC-2107 and PTC-2113, Perseus SENOLYTIX’s PTC-2109, PTC-2110, PTC-2117 and PTC-2128 and Ponce Aurora’s PTC-2108, PTC-2111 and PTC-2116 are all powered by MitoXcel Technology.
Focus on Eos SENOLYTIX
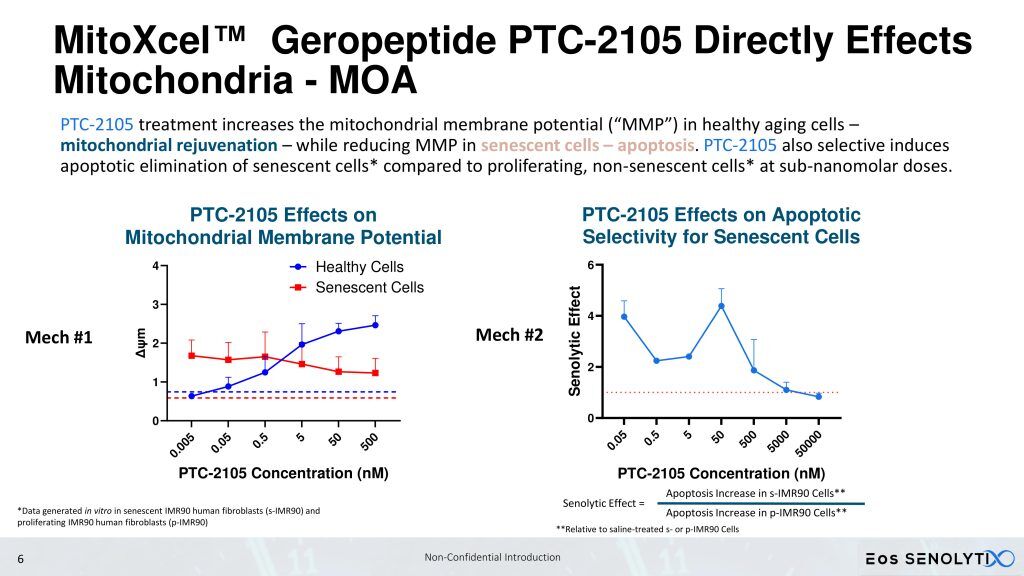
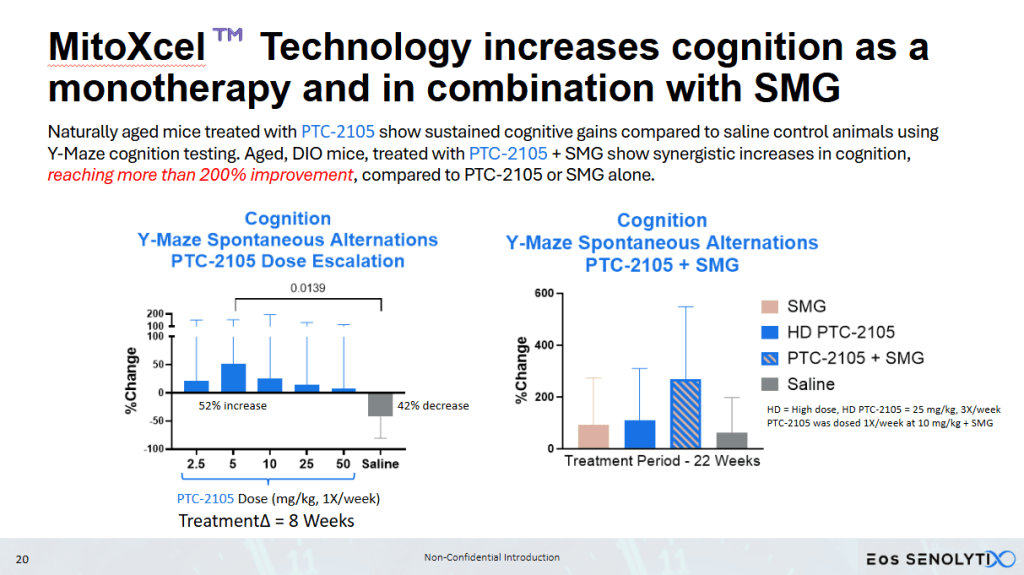
Eos SENOLYTIX is a longevity company using advanced technologies to develop novel therapeutics targeting the fundamental mechanisms of aging. Its lead programs, powered by MitoXcel Technology, have the remarkable ability to rejuvenate naturally aged mice via two separate mechanisms, both targeting the Mitochondrial Membrane Potential (MMP, ΔΨm), to dramatically eliminate senescent cells throughout the body in the medium to longer term, and to enhance the efficiency of mitochondrial function in aging cells, in the short term, with no apparent adverse side effects. Our MitoXcel geropeptides work via a completely novel mechanism of action, targeting the lower and declining ΔΨm found in senescent cells compared with non-senescent cells, and in aging cells in general, that develops as we age and that occurs in all living organisms. By targeting the fundamental processes driving aging and aging-related diseases, we have a unique opportunity to intervene in the aging process in ways that were once thought impossible.
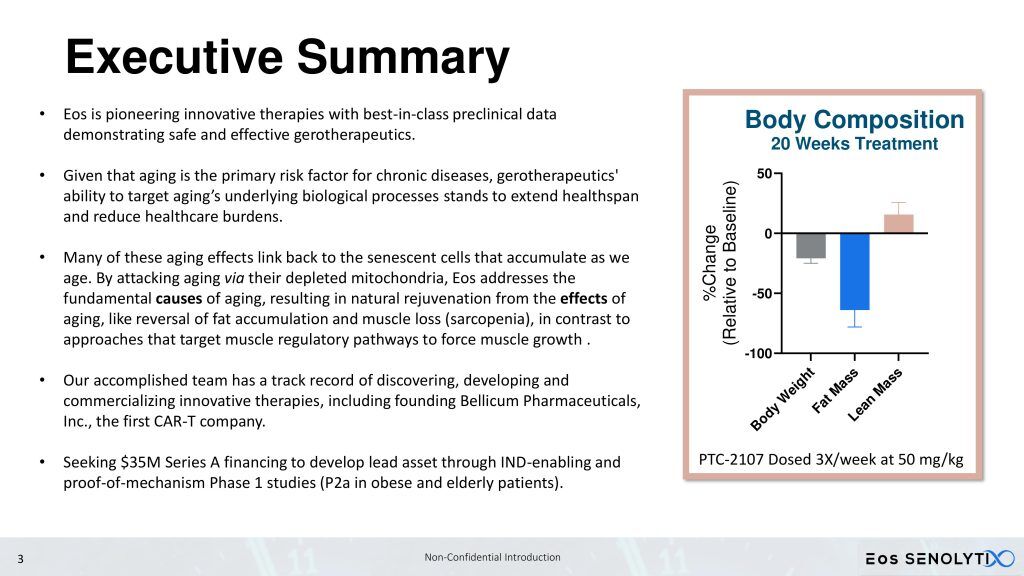
- Eos is pioneering innovative therapies with best-in-class preclinical data demonstrating safe and effective gerotherapeutics.
- Given that aging is the primary risk factor for chronic diseases, gerotherapeutics’ ability to target aging’s underlying biological processes stands to extend healthspan and reduce healthcare burdens.
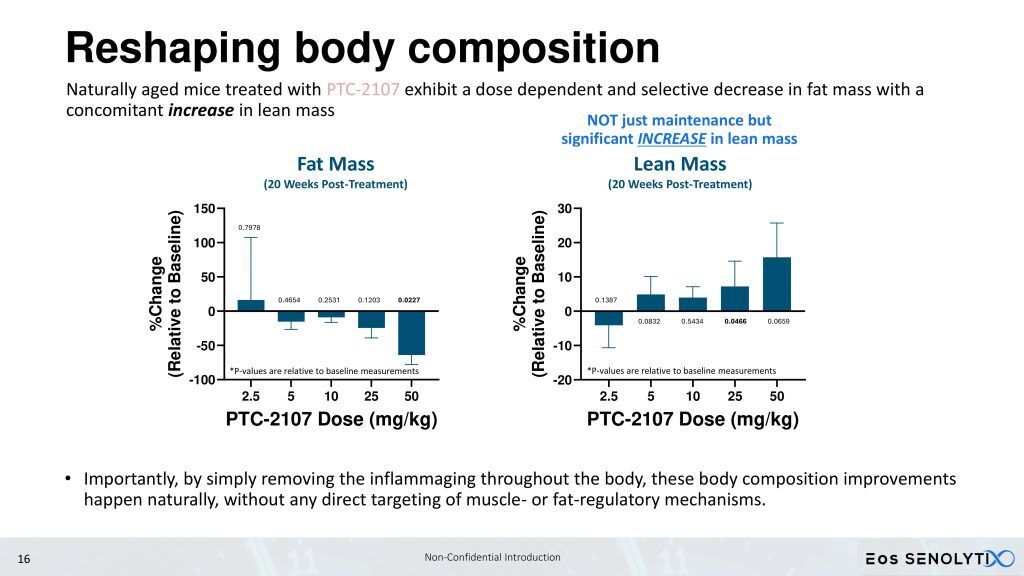
- Many of these aging effects link back to the senescent cells that accumulate as we age. By attacking aging via their depleted mitochondria, Eos addresses the fundamental causes of aging, resulting in natural rejuvenation from the effects of aging, like reversal of fat accumulation and muscle loss (sarcopenia), in contrast to approaches that target muscle regulatory pathways to force muscle growth.
- Our accomplished team has a track record of discovering, developing and commercializing innovative therapies, including founding Bellicum Pharmaceuticals, Inc, the first CAR-T company.
- Seeking $35 million Series A financing to develop our lead asset through IND-enabling and proof-of-mechanism Phase 1 studies (P2a in obese and elderly patients).
Eos SENOLYTIX’s novel proprietary geropeptides, the first aimed at an aging-specific target, have demonstrated remarkable ability to rejuvenate naturally aged mice via two separate mechanisms, firstly, by targeting the Mitochondrial Membrane Potential (MMP, ΔΨm) to efficiently eliminate senescent cells throughout the body, and secondly, by enhancing the efficiency of mitochondrial function in aging, non-senescent cells, with no apparent adverse side effects. Our peptides work via a completely novel mechanism of action, targeting the lower and declining ΔΨm that develops as we age [1].
Our candidate gerotherapeutics address both the inflammaging driving reduced healthspan due to increasing burden of senescent cells and the reduction in mitochondrial efficiency in non-senescent cells as we age, via a single, aging-specific target, the ΔΨm [2,3].
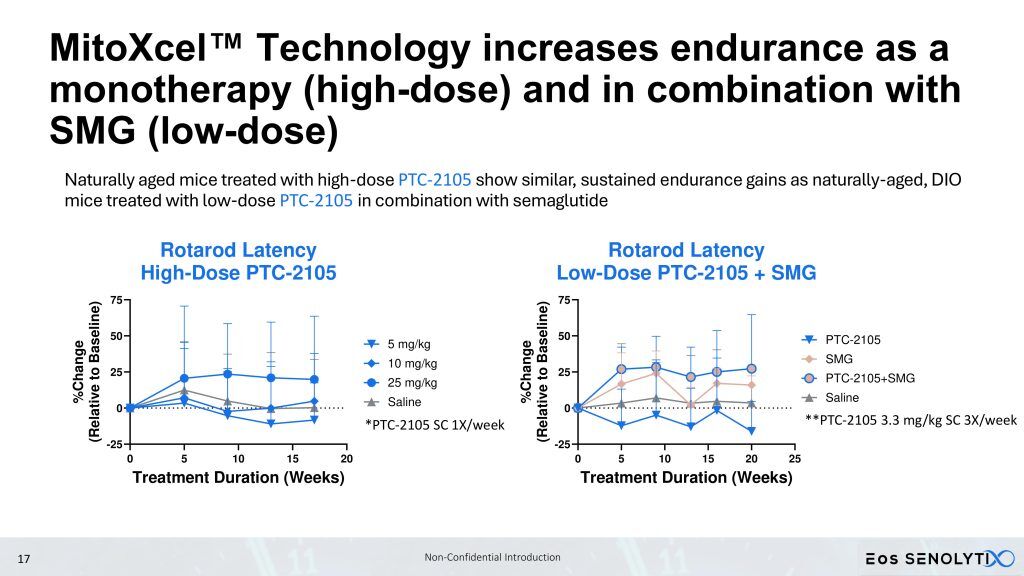
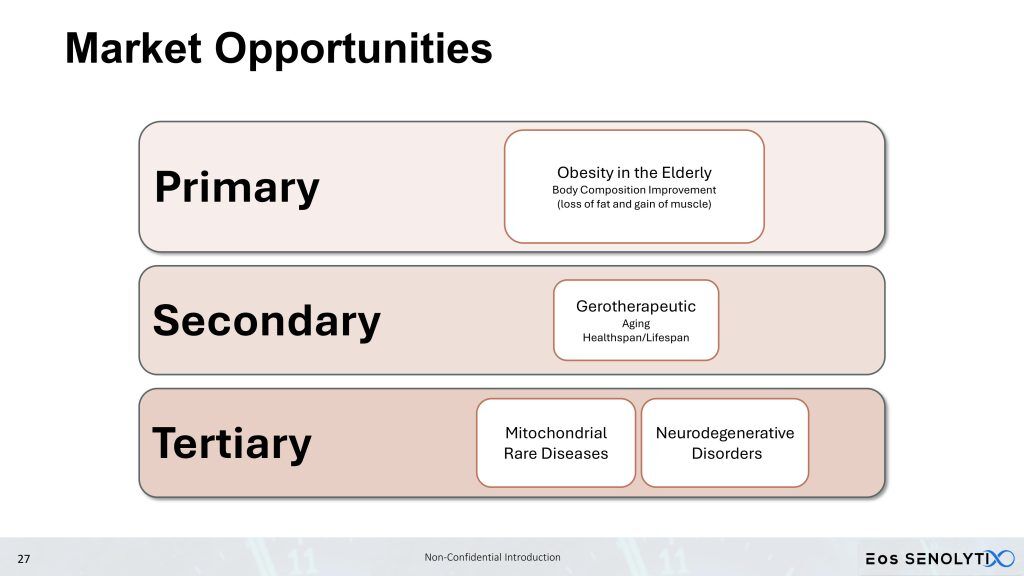
We are progressing towards performing a Phase 1b/2a clinical trial with our lead clinical candidate geropeptide, PTC-2105, in elderly obese patients, including sarcopenia and sarcopenic obesity. Sarcopenia is defined as the age-related progressive loss of muscle mass, strength, and function. By targeting and inducing apoptosis in senescent cells, as well as increasing mitochondrial efficiency in non-senescent cells, PTC-2105 aims to reverse sarcopenia, reducing fat while building muscle and bone mass, in patients with sarcopenic obesity, increasing strength and function, while simultaneously improving healthspan and reducing the burden of age-related diseases, fulfilling the promise of a true gerotherapeutic.
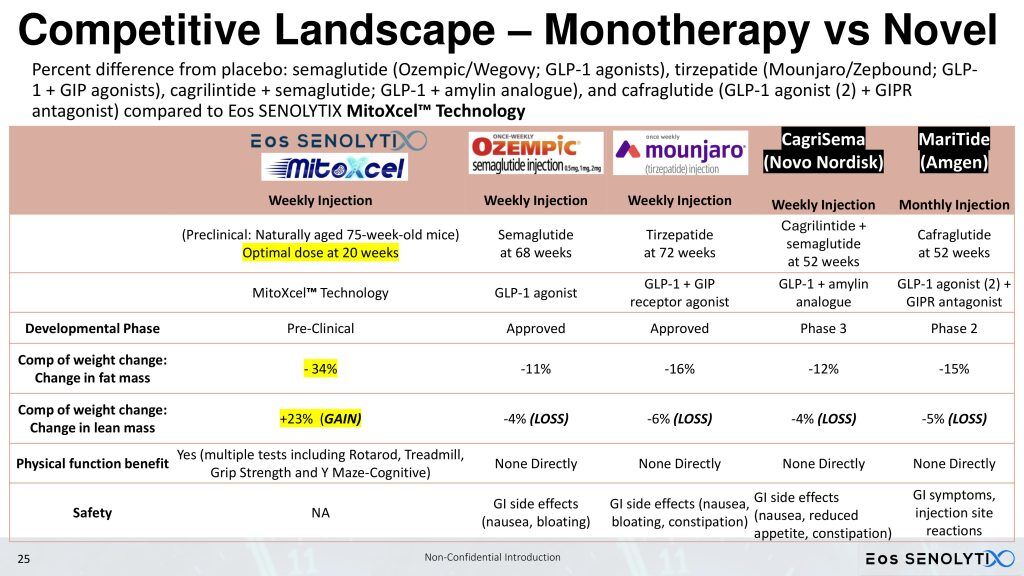
Sarcopenic patients, in particular, are not candidates for GLP-1 agonists, which reduce lean body mass, something which is already critically low in these patients. In preclinical mouse studies, PTC-2105 has demonstrated the ability to improve both body composition by DEXA scan, reducing fat but increasing lean body and bone mass, and healthspan as measured by Rotarod, motorized treadmill, forelimb grip strength, and Y maze, which are mouse correlates to measures in the human Intrinsic Capacity (IC) score [4,5,6], in both male and female C57BL/6 mice.
Our lead clinical candidate geropeptide, PTC-2105, selectively and safely eliminates a majority of the senescent cells found in every organ of the body, which are known to contribute to aging and age-related diseases. Senescent cells accumulate with age, leading to chronic inflammation, tissue dysfunction, and the progression of various age-related conditions. The elimination of senescent cells has been shown to improve tissue function and extend healthspan and lifespan in preclinical mouse models [7,8,9,10,11,12,13].
Focus on Phoenix SENOLYTIX
Phoenix SENOLYTIX is a longevity company using advanced technologies to develop novel gene therapies targeting the fundamental mechanisms of aging. Its lead programs are based on its two novel gene therapy platforms, ApoptiCIDe-CE, our purposeful cell elimination gene therapy platform, and ApoptiCIDe-RGT, our regulated gene therapy platform. Both use our proprietary enhanced, cell elimination switch technology that we originally developed as CaspaCIDe,as founders of Bellicum Pharmaceuticals, for use in T cells.
CaspaCIDe remains the world’s most used, clinically validated cell therapy safety switch, designed to be used only sporadically in the advent of a severe uncontrolled cell therapy toxicity. It has successfully been used to control toxicities in CAR cell therapy-treated patients experiencing CRS and the difficult-to-manage ICANS ~ 10 to 12 times in different settings by different investigators with 100% success rate. ApoptiCIDe is our re-imagined, proprietary version of CaspaCIDe™ that uses the same iCasp9 rimiducid-triggered switch as CaspaCIDe but replaces Bellicum’s intravenous rimiducid formulation with Phoenix’s proprietary SQ/IM rimiducid formulation as its wholly owned small molecule dimer ‘trigger’ for ApoptiCIDe.
Rimiducid was formulated by Bellicum Pharmaceuticals, Inc, for use with their cell therapy safety switch, CaspaCIDe, under their IND, as a 40-mg infusion (5 mg/ml x 8 mL (25% Solutol HS15) into 100 mL saline), administered over 2 hours in a monitored infusion center. This IV infusion remains cumbersome and costly, and its excipient, Solutol HS15, itself, is associated with infusion reactions. Phoenix’s proprietary, reformulated rimiducid, PTC-1202, is convenient as either a simple and quick intramuscular (IM) or subcutaneous (SC) injection and may potentially be further formulated into an autoinjector pen format.
On our ApoptiCIDe-CE purposeful cell elimination gene therapy platform, our lead products include ApoptiCIDe-CE-GERO-001 and ApoptiCIDe-CE-GERO-002 targeting senescent cells throughout the body for purposeful cell elimination for aging related disorders and ApoptiCIDe-CE-WAT-001, targeting adipocytes for purposeful cell elimination as a more immediate and effective method of eliminating fat without affecting lean body or bone mass for patients with morbid- or sarcopenic-obesity.
Using our ApoptiCIDe-RGT regulated gene therapy platform, we have developed ApoptiCIDe-RGT-Klotho as the prototype Proof of Concept for this platform, that demonstrates our ability to reduce or eliminate the production of a gene-therapy delivered-protein or -antibody therapeutic within hours of administration of PTC-1202. This safer, regulated gene therapy platform technology is available for out-license. We are also developing ApoptiCIDe-RGT-MitoX-001, ApoptiCIDe-RGT-MitoX-002™and ApoptiCIDe-RGT-MitoX-003 that deliver our novel, proprietary MitoXcel technology in a regulated gene therapy format for age-related disorders and cancer.
What can be called a ‘senolytic’?
Field needs a more stringent classification
Scientific definition of senolytics
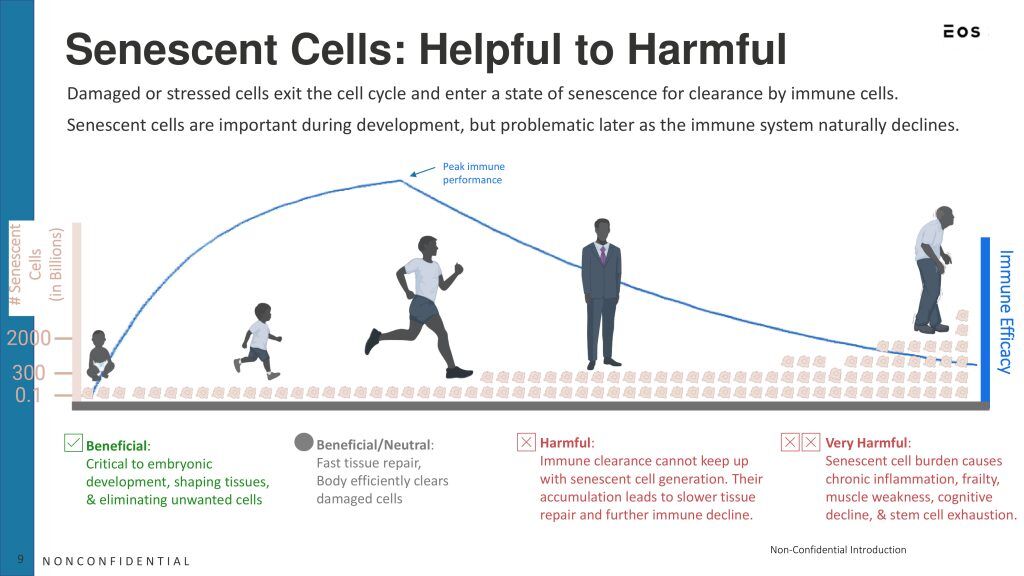
- Senolytics are broadly defined in the scientific literature as agents that selectively induce apoptosis in senescent cells – cells that have stopped dividing and contribute to aging and age-related diseases through the secretion of inflammatory factors known as the senescence-associated secretory phenotype (SASP).
- Key characteristics of senolytics:
- Target Senescent Cell Anti-Apoptotic Pathways (SCAPs) to overcome resistance to cell death.
- Aim to reduce the burden of senescent cells in tissues to improve function and delay aging-related decline.
- Include compounds like Dasatinib, Quercetin, Navitoclax, and Fisetin, among others.
Regulatory status (FDA & EMA)
- FDA: There are no senolytic drugs approved specifically for targeting senescent cells. While compounds like Dasatinib are FDA-approved for other indications (e.g., leukemia), their use as senolytics remains experimental.
- EMA: Similarly, the EMA provides general guidance on drug labeling and product information but does not recognize ‘senolytic’ as an official therapeutic class.
- Both agencies require:
- Demonstrated clinical efficacy and safety for the proposed indication.
- Clear mechanism of action, validated biomarkers, and reproducible outcomes.
- Standardized labeling and product information, but no mention of senolytics as a recognized category
Challenges to classification
- Heterogeneity of senescent cells across tissues and diseases complicates standardization.
- Lack of robust biomarkers for identifying and tracking senescent cells in vivo.
As a field, we need to be much more precise about what can be called a “senolytic therapy” based on two key criteria:
- What percent of senescent cells are demonstrably and reliably eliminated by the candidate therapeutic in acceptable pre-clinical models?
- How specific is the therapeutic in targeting senescent cells for elimination versus other biologic processes important in all cells generally?
Suggested “senolytic” criteria:
- Demonstrated ability to eliminate at minimum >50% of senescent cells in at least two organs of the body by assay of at least two senescent-cell biomarkers, e.g., SA-βgal, p16, p21, uPAR and/or Lamin B1.
- In addition, a candidate senolytic should have demonstrated specificity in targeting senescent cells over nonspecific effects on cellular pathways important in all cells, along with a reasonable Method of Action explaining that specificity.
Eos SENOLYTIX‘s lead MitoXcel clinical candidates, PTC-2105 and PTC-2107, eliminate ~60 to 80% of senescent cells in all organs of the body, including the brain, in over 20 well-controlled studies performed in naturally aged 75-week or 90-week-old mice, within 8 weeks of treatment as demonstrated and confirmed by all of the above-listed biomarkers of senescent cells. Furthermore, our proprietary MitoXcel Technology is highly specific at targeting the two types of cells that demonstrate a lower Mitochondrial Membrane Potential, aging senescent cells and cancer cells.
Phoenix SENOLYTIX’s lead senolytic clinical candidate, ApoptiCIDe-CE-GERO-002™ plus proprietary dimerizer activating agent PTC-2102, eliminated ~60 to 70% of senescent cells in the spleens of treated naturally aged 75-week-old mice as assayed by SA-β-gal and Lamin B1.
Key data








Graphics and photographs courtesy of SENOTHERAPEUTIX, Inc
[1] https://pubmed.ncbi.nlm.nih.gov/35775483/
[2] https://www.nature.com/articles/nature16932
[3] https://www.nature.com/articles/s43587-022-00340-7
[4] https://link.springer.com/article/10.1007/s11357-024-01226-9
[5] https://www.nature.com/articles/s41586-024-08026-3
[6] https://pubmed.ncbi.nlm.nih.gov/33211821/
[7] https://www.nature.com/articles/s41586-020-2403-9
[8] https://www.nature.com/articles/s43587-023-00560-5
[9] https://www.nature.com/articles/nature10600
[10] https://www.nature.com/articles/nm.4385
[11] https://onlinelibrary.wiley.com/doi/10.1111/acel.14340
[12] https://onlinelibrary.wiley.com/doi/10.1111/acel.13296
[13] https://onlinelibrary.wiley.com/doi/10.1111/acel.12950


