A new roadmap identifies 100 open problems and highlights where biogerontology still lacks clarity and cohesion.
The effort to understand human aging has never wanted for hypotheses; what it has often lacked is a shared map of the questions that most require attention. A new paper in GeroScience attempts to supply just that by presenting a curated list of 100 open problems in aging biology. Developed through community submissions, a structured workshop and an NLP-driven analysis of more than 200,000 PubMed abstracts, the list reflects both the field’s preoccupations and its blind spots. Led by Professor João Pedro de Magalhães and an international roster of coauthors, the work proposes a framework for prioritizing research in a discipline that is expanding rapidly, but not always coherently [1].
The roadmap spans fundamental questions about why aging occurs, mechanistic uncertainties across cellular and molecular pathways, translational concerns relating to biomarkers and interventions, and comparative gaps that continue to challenge cross-species inference. Some problems have been studied for decades; others are relatively new entrants shaped by recent advances in epigenetics, reprogramming and multi-omics. Together, they form an attempt to codify where biogerontology is today and where it might need to travel next.
Longevity.Technology: Biogerontology has always wrestled with the uncomfortable truth that progress depends as much on choosing the right questions as on answering them, yet the field’s questions have tended to sprawl – diffuse, sometimes redundant and occasionally shaped more by personal preference than collective strategy. This new catalog of 100 open problems attempts to bring a degree of discipline to the endeavor, offering not a tidy manifesto but a structured mirror held up to a discipline that is still defining its own contours. The mix of familiar puzzles and less-traveled avenues is telling; it reveals a community confident enough to acknowledge its blind spots and ambitious enough to gather them into a shared framework.
What emerges is less a list and more a provocation – a reminder that geroscience advances unevenly, pulled toward fashionable mechanisms even as entire territories remain curiously underexplored. The prominence of molecular pathways, interventions and biomarkers contrasts sharply with the sparse attention given to environmental factors or diversity in aging, as if the field has collectively agreed to tidy its laboratory bench while ignoring the rest of the room. Yet this self-awareness is precisely what makes the roadmap valuable. By combining expert insight with the impartiality of NLP-driven analysis, it challenges researchers to re-examine their assumptions and funders to broaden their horizons. Whether it succeeds will depend not on its length but on whether the community chooses to treat it as a living document rather than a decorative one.
How the list was built
The authors describe the process as a “collective and systematic effort,” drawing on 290 initial submissions, later refined to 204 after removing duplications and non-relevant entries. These were analyzed with PubMedBERT and MedCPT to quantify how frequently each problem appeared in the literature – a pragmatic way of revealing both entrenched research interests and areas that have received little attention. Problems ranged from having just a single associated PubMed article to more than ten thousand, showing stark imbalances in effort [1].
As the authors note, “the representation of individual open problems ranged from 1 to 10,808 articles,” which highlights how unevenly the field distributes its intellectual energy [1]. Some questions, such as “Why do we age?” and “Does somatic mutation accumulation cause aging?”, dominate entire swathes of the literature. Others – often related to underexplored mechanisms or experimental approaches – remain scarcely addressed.
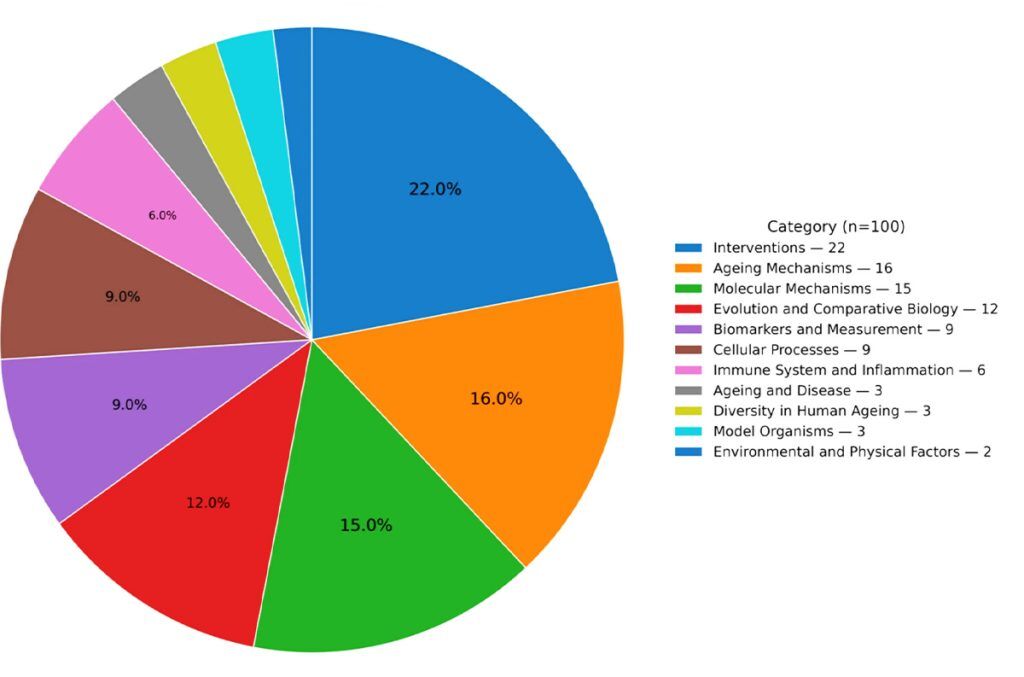
The resulting 100 problems were organized into 11 themes. Aging mechanisms, molecular mechanisms and interventions occupy the largest slices of the pie chart presented in the paper; environmental influences, diversity in human aging and organ-specific aging are notably smaller categories. This distribution may reflect scientific priorities, but also structural biases in the broader research ecosystem.
Pie Chart of the total number of open problems by catagory
Recurring questions and evolving debates
Several of the “top” problems will be familiar to anyone following aging science. The field continues to grapple with whether there are fundamental, conserved aging processes and how these interface with age-related disease. Mitochondrial dysfunction, cell senescence, immune dysregulation and genomic instability feature prominently, as do questions about the pace of aging and the determinants of species longevity.
The translational strand includes open debates around biomarker validity – a topic of increasing urgency given the need for reliable measures in trials of gerotherapeutics. One listed question asks: “How can we measure intrinsic biological age in individuals and translate this knowledge into accurate biomarkers of aging?” Another probes how interventions should be prioritized for clinical trials, pointing toward the regulatory and evidentiary bottlenecks that still constrain progress.
Notably, the roadmap also incorporates questions that modernize the classic puzzles articulated by Bernard Strehler nearly 50 years ago. The authors highlight that while many themes remain constant – genetics, comparative biology, transcriptional changes – new priorities such as senolytics, partial reprogramming and large-scale omics reflect the tools and ambitions of contemporary geroscience [1].

Where the gaps lie
The paper is not shy about pointing to areas where research is lacking. Environmental and physical factors occupy only 2% of the problems, despite growing evidence that exposures and lifestyle may interact with biological aging pathways in complex ways. Diversity in human aging is similarly underrepresented, raising questions about how well current models and biomarkers generalize across populations [1].
The authors concede that their list “will still reflect biases in how contemporary scientists perceive aging and those topics they find more important. [1]” Yet the transparency of the method – particularly the NLP-driven prevalence analysis – helps reveal these biases rather than conceal them. Whether the field chooses to redirect attention accordingly remains to be seen.
A wider lens on priorities
By placing these problems on an interactive platform, the Longevity Knowledge App, the group aims to keep the list dynamic. Users can explore each problem, examine metadata and contribute proposals for tackling them. This approach treats the roadmap not as a static artifact but as infrastructure – a scaffold for discussion, collaboration and course-correction as the field expands.
A question of future direction
If aging science is indeed reaching a pivotal moment, as many argue, then articulating the uncertainties that persist is a necessary step toward shaping its trajectory. The most valuable outcome of this roadmap may not be the questions themselves, but the collective acknowledgment that progress requires shared priorities as well as shared ambition.
We’ll be speaking to Professor de Magalhães about this paper, what it means for geroscience and how the industry should react later this week – stay tuned!
[1] https://link.springer.com/article/10.1007/s11357-025-01964-4


