Tune Therapeutics’ targeted epigenetic editing approach is already in the clinic as company targets multiple opportunities in healthspan.
Last month we brought you news of Tune Therapeutics, which landed a whopping $175 million funding round for its “epigenetic tuning” approach to treating chronic diseases. Backed by the likes of global non-profit Hevolution Foundation, and with a stated goal to improve human healthspan, the company is taking a unique approach that brings genetic medicine technologies closer to the world of epigenetic reprogramming.
Longevity.Technology: Epigenetic changes, the modifications to our DNA that regulate whether certain genes are turned on or off, accumulate with age, leading to reduced cellular resilience, immune function decline and increased susceptibility to disease. Ever since Professor Shinya Yamanaka made the Nobel Prize winning discovery that an adult cell could be reverted back into an embryonic stem cell by exposing it to certain transcription factors, others have sought to explore epigenetic reprogramming’s potential to benefit human health. A host of well-funded companies have since been founded to harness its power, however, we have yet to see an epigenetic reprogramming therapy enter human trials.
While Tune operates in the same ballpark, the company’s approach is vastly different, and is already engaged in a Phase trial of its technology – an epigenetic silencer for chronic Hepatitis B. We sat down with Tune CSO Derek Jantz to find out more about what sets the company apart.
According to Jantz, Tune’s approach lies about halfway between gene editing and Yamanaka-style epigenetic reprogramming. Essentially, the company’s platform, called Tempo, provides a way of achieving epigenetic reprogramming in a more targeted and intentional manner.
Precise control of gene expression
Epigenetic reprogramming by Yamanaka factors presents a host of challenges that have to be addressed, from safety concerns over activating oncogenes that could lead to cancer, to the technical hurdle of effectively delivering transcription factors to the right cells and tissues in the body.
“What we’re able to do with our platform is go into a cell and very precisely control the expression of certain genes through epigenetics,” explains Jantz. “We have this ability to turn genes on or turn genes off, turn them up or down a little bit. We can control exactly how much we’re changing – the amount of a specific protein that’s getting made in a given cell – and also how long that effect lasts.”
This last point is important and addresses another concern when it comes to other reprogramming approaches, in that it can be difficult with transcription factors to precisely control the duration of epigenetic changes, with the risk of effects being either too transient or too permanent.
“For a lot of the kind of changes that are being considered for epigenetic control, you may not want something that lasts for life,” explains Jantz. “You may want something that lasts for three weeks or for six weeks. We have a platform that really allows us to do that in a very precise way, so a little bit more targeted, and less haphazard than some of the epigenetic reprogramming approaches that others are taking.”
While some in the cellular reprogramming field use terms along the lines of “making cells younger,” Jantz is keen to stay away from such language.
“I think saying you can make cells ‘young’ is over promising – nobody can do that today,” he says. “In fact, I think that language is going to do far more harm than good, because I worry that it’s setting expectations at a level that can’t practically be achieved.
“What we can do – in a very precise way – is to reawaken mechanisms that are present in younger cells but are turned off in older cells. But we’re not making the cells young, we’re making it look a little bit more like a healthy cell.”
CRISPR-powered approach
The company’s primary tool – something Jantz calls an “a genetic tuning platform” – is conceptually similar to CRISPR-Cas or nuclease-based gene-editing approaches, yet it differs in several critical ways. One of the limitations of gene editing is that it is permanent and irreversible, which makes it well suited to knocking out single-copy disease genes, but ill-suited to fine-tuning subtle “networks” of genes that act in concert to orchestrate cellular health and development.
“Gene editing is quite binary in the sense that you can knock a gene out, but you’re taking it out forever because you’re actually changing the underlying DNA sequence – you’re fundamentally changing the DNA,” explains Jantz. “But that’s not how biology works – particularly when you think about longevity and healthspan. Your DNA (or genetics) doesn’t change that much as you grow older. What does change is your epigenetics, and the patterns of genes that are being turned on and off in response to whatever it is that has programmed our bodies to break down over time.”
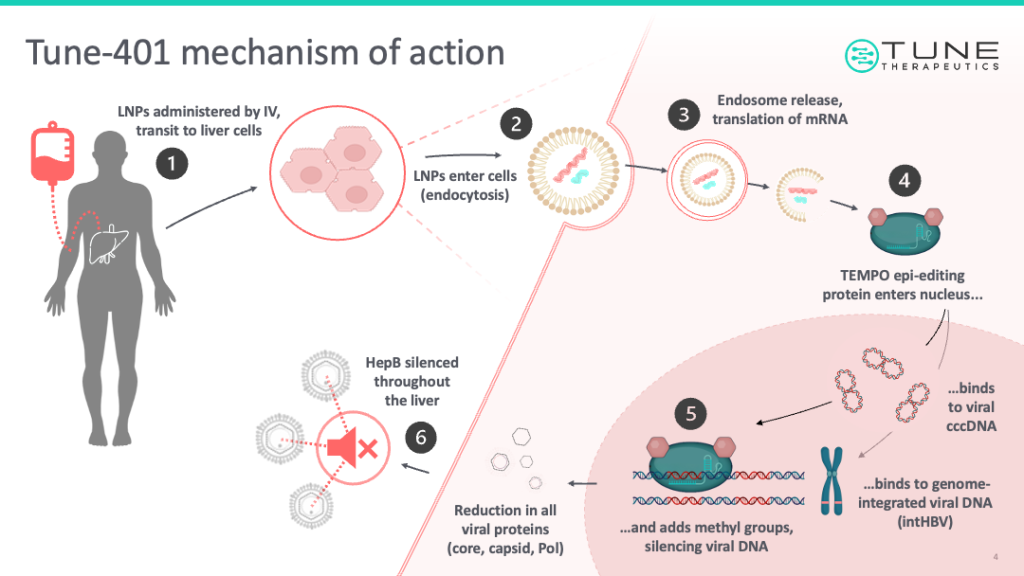
Tune’s approach involves the use of a “catalytically dead” CRISPR-Cas DNA-binding protein, which binds to DNA but does not (indeed cannot) cut it. Rather this dCas (or “dead Cas”) protein is tethered to one or more “effector” proteins, which add or remove epigenetics marks that render targeted genes active or inactive without altering their DNA sequence in any way.
To get these epi-editing constructs into the body, Tune encodes both proteins (along with a DNA-targeting “guide” RNA) into custom-built RNA molecules, wrapped in a lipid nanoparticle shell. The LNP is designed to deliver the RNA to a specific tissue type, such as the liver. There, the RNA is taken into cells and translated into epi-editing constructs – which in turn pinpoint the desired genes, and dial them up or down for the desired therapeutic effect.
“The level of precision and control offered by this system allows us to think about very specific applications, very specific diseases that are associated with aging,” says Jantz. “Think about your body’s natural ability to regenerate when you were a kid: those mechanisms are still there, they’ve just been turned off or turned down a little bit. And what we’re doing is turning them back on, and restoring your body’s ability to regenerate in very precise ways that manifest in very distinct disease pathologies.”
‘Unlimited potential’
While Jantz believes the technology has “almost unlimited potential,” Tune’s first clinical trial targets chronic Hepatitis B – a virus that infects the liver and is the primary cause of liver cancer worldwide.
“One of the ways that your body tries to deal with virus infections like Hepatitis B is it tries to use epigenetic control to turn the virus off,” explains Jantz. “But the virus has evolved ways to evade the machinery that your body has to epigenetically silence it. They’ve got all these defense mechanisms built in, but with our technology, we can get through the defenses and we can turn the virus off in such a way that we think it won’t come back on.”
“It’s a great starting place for us. We had to pick something to start with as a company that we can actually turn into a drug that we can put into people, and show that, yes, in fact, this technology is a fundamentally new therapeutic modality.”
That Tune is already in clinical trials is remarkable in itself, and Jantz believes the concept of epigenetic editing is more palatable to many than gene editing.
“I spent 16 years developing gene editing therapies and, having talked to regulators and health agencies and patients and investigators, I will say that it is a much, much easier path with an epigenetic editing therapy that that doesn’t change the underlying DNA sequence,” he says. “Regulators are far more comfortable with it. Investigators are more comfortable with it. Patients seem to be a lot more comfortable with this. So it’s been fairly straightforward. In fact, I’d say remarkably straightforward to get this into the clinic.”
Multiple future opportunities
Looking beyond Tune’s first trial, Jantz says the company is exploring multiple potential applications of its technology to improve human healthspan. One of those areas is regenerative medicine.
“We’re very interested in this concept of cellular reprogramming and taking one cell type that may be healthy or unhealthy and turning it into a different cell type that is healthy and can be used in a regenerative medicine or tissue engineering,” he says. “For example, can we potentially take a skin or blood cell from a person, and use our technology to turn that cell into a stem cell, and then use the technology again to turn that stem cell into something else, a hepatocyte, maybe; a beta-islet cell for diabetes; or blood cells for oncology or autoimmune disease. There’s a lot that we could do there.”
Jantz also suggests the possibility of targeting cholesterol control. Tune has already demonstrated the durable repression of the PCSK9 gene in non-human primates, resulting in a sustained reduction of LDL cholesterol levels nearly two years after a single treatment.
“That’s a great example of a very specific indication for a technology like ours,” he says. “We’ve already shown it works in non-human primates, and by the way, the composition of that drug is remarkably similar to the composition of the Hepatitis B drug that’s currently in the clinic. Though the indications are very different, on the molecular level the fundamental epi-editing constructs are very similar to one another. So you might expect that it would have a similar path to getting to the clinical trial stage, if we were to decide to move it forward.”
Allowing himself a moment to think about the far distant future, Jantz says he can envisage a time where the technology extends beyond chronic disease indications into broader healthspan applications.
“As we identify these common mechanisms that are in place across the body that are involved in the epigenetic manifestations of aging, can we start to find and, hopefully, a reasonable number of epigenetic modifiers that can address multiple different aspects of aging in one shot?” he says. “ I think that that is certainly doable, and we’re aiming to get to a point where we can do this on a much larger scale.”


