Eos SENOLYTIX Founder Dr Kevin Slawin unveils the groundbreaking science behind proprietary peptides that target aging at its core.
Eos SENOLYTIX is a longevity company using advanced technologies to develop novel therapeutics targeting the fundamental mechanisms of aging. Its lead programs, PTC-2105 and PTC-2107, both proprietary gerotherapeutic peptides, have demonstrated the ability to rejuvenate naturally aged mice via two separate mechanisms, both targeting the mitochondrial membrane potential (MMP), also called the “Δψm” (pronounced “delta psi em”), the difference in electrical potential between the mitochondrial matrix and the cytosol generated as protons are pumped out during the process of oxidative phosphorylation, during which the mitochondria “burn” glucose into ATP molecules, the packets of energy used by cells to fuel all of life’s processes, in the presence of oxygen.
The company’s peptides work via a novel mechanism of action; they target the lower and declining MMP (“Δψm”) found in senescent cells which develops as we age, that reduces mitochondrial function and ATP production, a universal finding across all living species as they age. By targeting this single, aging-specific target, the peptides have demonstrated the ability to rejuvenate naturally aged mice via two separate mechanisms: (1) to steadily and profoundly eliminate senescent cells throughout the body in the medium to longer term, and (2) to enhance the efficiency of mitochondrial function in those senescent cells in the short term, with no apparent adverse side effects observed. By targeting the fundamental processes driving aging and aging-related diseases, Eos SENOLYTIX claims to have a “unique opportunity to intervene in the aging process in ways that were once thought impossible, to improve healthspan and increase lifespan”.
Having just presented exciting data at Founders Longevity Forum in Singapore, Founder and CEO, Kevin Slawin, MD takes us on a deep dive into the origins of Eos SENOLYTIX, Inc and the science behind its proprietary gerotherapeutic peptides, exploring their novel MOA and the results in pre-clinical models that they have developed. Plus, register to view and download the Eos SENOLYTIX deck presented by Dr Slawin earlier today.

In the beginning
After our early belief in immunotherapy for cancer, which was at odds with the then current mainstream thought, led us to found Bellicum Pharmaceuticals, the first CAR T cell company deploying controllable cellular immunotherapy, in 2004, and which we took public on the NASDAQ in a 2014 $160 million IPO at a $500 million market cap after the world caught up to us, we took notice that Jan van Deursen’s group of researchers at Mayo Clinic in Rochester, MN, using Bellicum’s CaspaCIDe™ inducible caspase cell “suicide switch” technology to create their p16Ink4a-ATTAC transgenic mouse model, had demonstrated dramatic improvements in healthspan and lifespan with no adverse events in this mouse model simply by eliminating senescent cells using our technology (Baker et al, 2011 and 2016). Two years after exiting Bellicum in 2017, I founded SENOTHERAPEUTIX, Inc. working in Houston, TX to develop a gene therapy targeting senescent cells for purposeful elimination based on that technology, which we improved and renamed ApoptiCIDe™.
My strong appetite for tackling the most important and challenging problems in medicine, that began my journey into the fields of geroscience and longevity medicine, now over 5 years ago, has led me to spin out our dramatic discoveries and innovative technologies into three wholly owned subsidiaries of SENOTHERAPEUTIX, Inc: (1) Eos SENOLYTIX, a longevity company developing novel gerotherapeutic peptides to improve healthspan and increase lifespan (2) Perseus SENOLYTIX, an oncology company developing novel gerotherapeutic peptides to target cancer via this same novel mechanism as a potentially safe and effective new class of cancer therapeutics and (3) Phoenix SENOLYTIX, developing novel, controllable gene therapies using our proprietary ApoptiCIDe™ purposeful cell elimination technology to improve healthspan and increase lifespan.
Chance favors the prepared mind
On one of my monthly trips to our Houston R&D facilities at K2 Bio in Houston, TX, from my home in Miami in November 2023, as my team was presenting to me the latest data updates to our experiments in progress, I realized that we had stumbled upon a new class of peptides that could safely and effectively eliminate senescent cells in every organ of the body. Our team set out on an ambitious and systematic program to broaden this discovery and determine the mechanism of action of these peptides, that we referred to as “senolytic”, because we learned that they effectively eliminated senescent cells. More recently, however, we made an astonishing observation in one of our studies that tipped us off to a second, equally important MoA for these peptides, that have led us to now refer to them as “gerotherapeutics”, since their senolytic activity is only half of this remarkable story.
Don’t overthink aging
One thing that led to my success in founding Bellicum Pharmaceuticals in the cellular immunotherapy field, was my strong belief that the body has a remarkable ability to heal itself – one of the basic principles of immunotherapy, in which one genetically modifies the immune system to target and eradicate cancer. While we were the first CAR T cell company, there are now hundreds of them because of the unbridled success of this approach.
At Eos SENOLYTIX, Inc, our founding belief has always been that one of the primary drivers of aging is the worsening systemic inflammation associated with aging, known as “inflammaging”. Inflammaging is caused by the senescence-associated secretory phenotype, or SASP, produced by the increasing senescent cell load accumulating in the body with age. If this hypothesis is correct, then simply by eliminating senescent cells, one should see a drop in systemic levels of the inflammatory SASP, which should then directly allow the body to then heal itself from the damaging effects of inflammaging, remodelling organs and improving their function, thus reversing the aging phenotype without further interventions.
In fact, there is already a plethora of proof-of-concept data generated in the p16Ink4a-ATTAC transgenic mouse model that this rejuvenatory process occurs once senescence-associated inflammaging is eliminated from the body (Ogrodnik et al, 2020, Palmer et al, 2019, Farr et al, 2017, and others). Furthermore, I believe it is almost futile to attempt to arrest or reverse the aging phenotype by any other methods without first addressing inflammaging. It would be like trying to row a boat upstream against a strong current. And in fact, data generated in the p16Ink4a-ATTAC transgenic mouse model has clearly established that once one removes the inflammatory environment in the body, many of the aging-associated pathologic phenotypes will heal themselves without further intervention.
Defining the problem: the senotherapeutic hypothesis
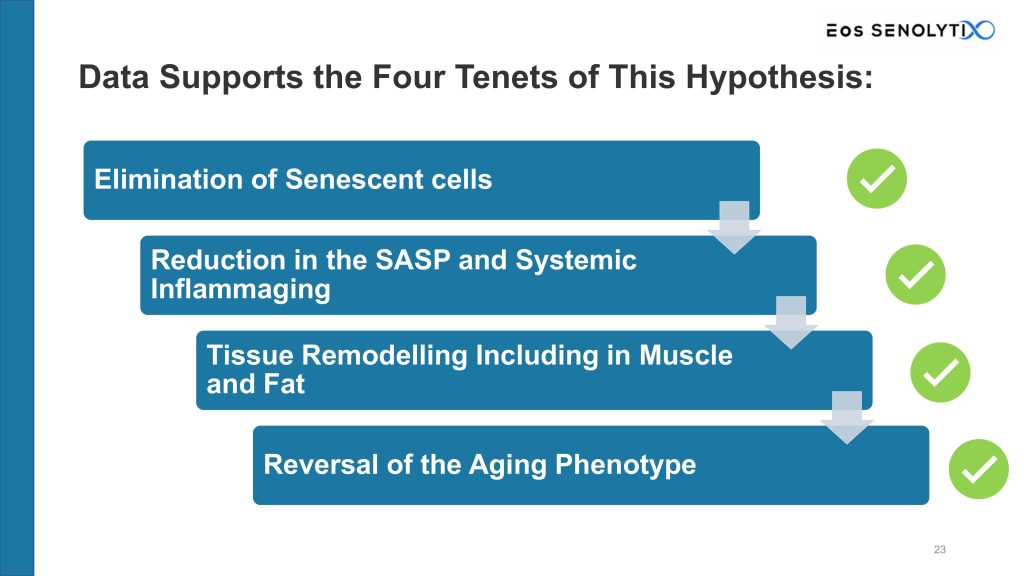
According to the Senotherapeutic Hypothesis, aging leads to systemic inflammation (‘inflammaging’) driven by senescent cells. Inflammaging is caused by the senescence-associated secretory phenotype, or SASP, produced by the increasing senescent cell load accumulating in the body with age. Inflammaging impairs organ function and causes aging and aging-associated diseases, e.g. cognitive decline, diabetes, heart failure, osteoporosis, etc.
If this hypothesis is correct, then:
- Elimination of senescent cells should lead to
- Reduction in the SASP and systemic inflammaging, which should then lead to
- Tissue remodelling, including in muscle and fat, which should then lead to
- Reversal of the aging phenotype without the need for further interventions.
Our data supports these four tenets of the senotherapeutic hypothesis
Eos’s gerotherapeutic peptides, PTC-2101, and clinical candidates PTC-2105 and PTC-2107 are 18- to 30-amino acids in length, that cross the blood brain barrier and are administered via SC injection over an 8-16 week course of treatment.
Key findings:
- Selective elimination of senescent cells in all organs of the body.
- Reduced inflammation – a significant decrease in SASP biomarkers.
- Tissue remodeling to a healthier phenotype in muscle and fat – improved body composition, including a reduction in fat but an increase in lean body and bone mass.
- Improved physical health – enhanced metabolism, muscle strength, and exercise endurance.
- Cognitive benefits – improved memory and motor coordination.
- Dose response curve demonstrated.
- Safety profile – normal liver enzyme levels and no adverse effects observed.
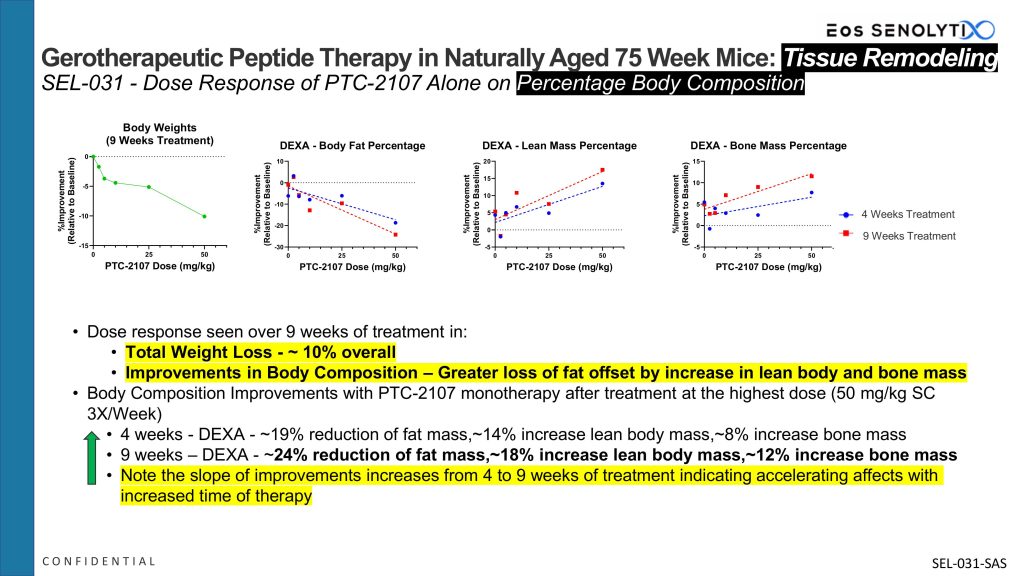
The above figure shows the effects on percentage body composition via tissue remodelling that occurs after 9 weeks of therapy with Eos’s clinical candidate gerotherapeutic peptide, PTC-2107, in a standard dose escalation study. Note the improved body composition after 9 weeks of therapy, including a 24% reduction in fat mass, an 18% increase in lean body mass and a 12% increase in bone mass, at the highest dose. Also note the strong dose response correlation with improvement in all three parameters of body composition.
Loss of lean body and bone mass has been the Achilles’ Heel of current GLP-1/GIP antagonists.
Furthermore, this improvement in body composition accelerates with time with continued treatment with PTC-2107.
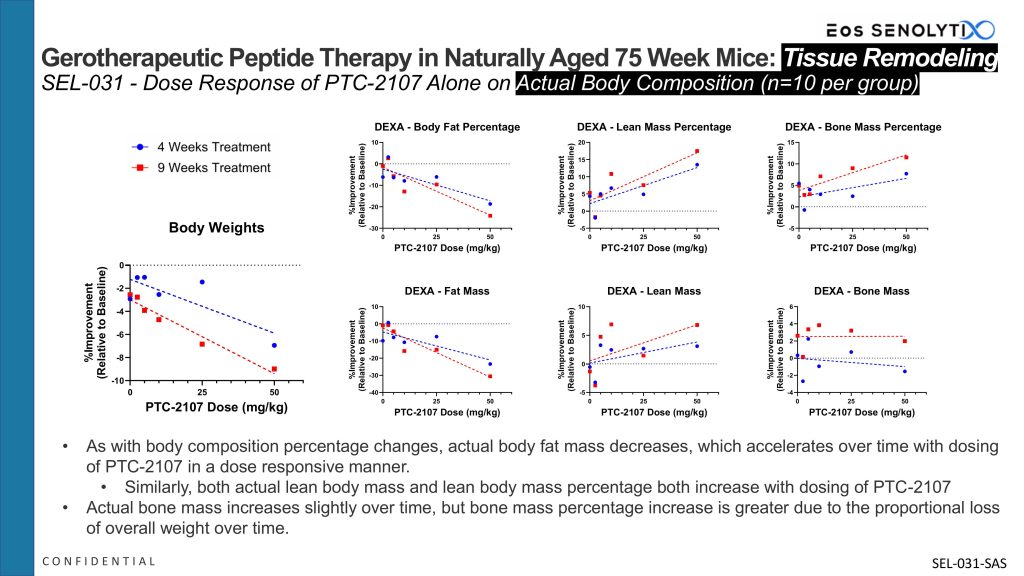
In the same study, when analyzing actual versus percentage body composition changes, actual body fat mass decreases (-31% at 9 weeks at the highest dose, p=0.04), which accelerates over time, as does body fat percentage, with dosing of PTC-2107 in a dose responsive manner. Similarly, both actual lean body mass (+6.8% at 9 weeks at the highest dose, p=0.09) and lean body mass percentage both increase over time with dosing of PTC-2107. Actual bone mass increases slightly over time, but bone mass percentage increase is greater due to the proportional loss of overall weight over time.
Register to view the full deck that includes this data and all the additional data supporting our key findings (see form at end).
Eos’s gerotherapeutic peptides: novel mechanism of action
Mechanism #1: Elimination of senescent cells via mitochondrial-mediated apoptosis
As humans age, mitochondrial function declines, leading to reduced ATP production efficiency and increased oxidative stress. This is a universal finding across all living species. The reduction in mitochondrial efficiency with age is in part due to protons bypassing the ATP synthase by “leaking” back across the inner mitochondrial membrane (IMM), which develops damage and dysfunction with age, short-circuiting the coupling of ATP synthesis and O2 consumption. This leak dissipates the MMP (Δψm) and reduces the efficiency of oxidative phosphorylation. The increased proton leak means that more oxygen is consumed to produce the same amount of ATP, reducing the overall efficiency of oxidative phosphorylation. The inefficiency leads to higher production of reactive oxygen species (ROS), which can further damage mitochondrial components and exacerbate the decline in function. This change in the cellular energetics is a sign that the ability of the mitochondria to meet the energetic demands of the cell is compromised. In addition to this general decline in mitochondrial efficiency with age, senescent cells have a lower MMP (Δψm) than non-senescent cells. In senescent cells, our gerotherapeutic peptides selectively trigger apoptosis via a direct effect on the Mitochondrial Membrane Potential (MMP) (ΔΨm), exposing them to prolonged mitochondrial transition pore opening and permeabilization of these mitochondria, leading to apoptosis and selective elimination of these cells. Our data suggests that this takes at least 8 weeks or greater of therapy to occur.

Astounding new data reveals a second mechanism at play
Because of these unique results in body composition improvement with treatment with our gerotherapeutic peptides, the first therapeutics that can convincingly and significantly reduce fat mass while increasing lean body and bone mass, we began an experiment to test our gerotherapeutic peptides alone and in various combinations with semaglutide, in 60-week-old mice fed a 60% high fat diet (Diet Induced Obesity or “DIO” Model). The results were astonishing:
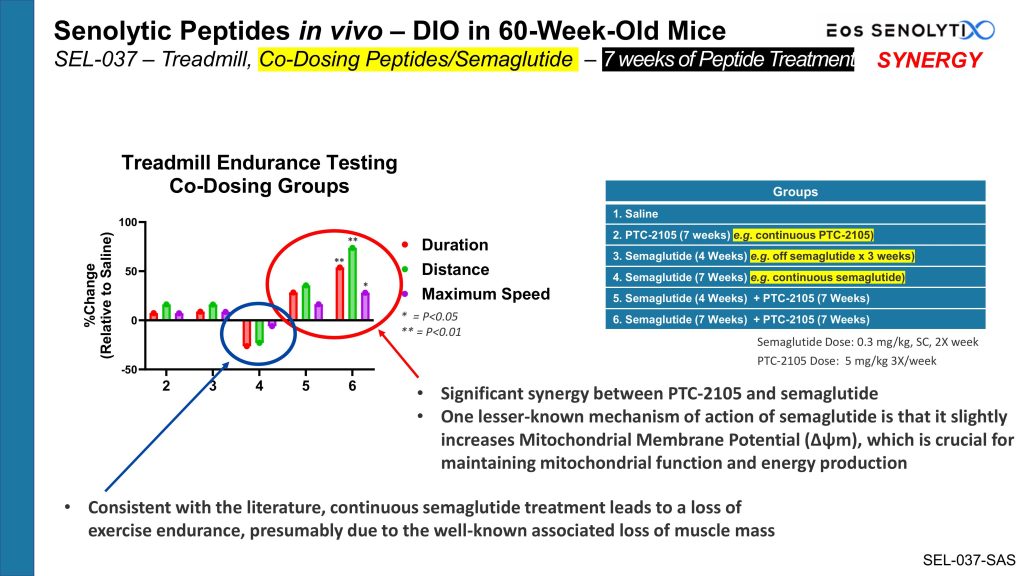
| Eos’s gerotherapeutic peptides work synergistically with semaglutide, acting at the Inner Mitochondrial Membrane to increase the MMP (Δψm) that decreases with age in all living organisms, dramatically improving exercise capacity, thus reversing the well-known decline seen with semaglutide treatment alone. When co-administered with semaglutide, clinical candidate gerotherapeutic peptide PTC-2105 dramatically improved exercise capacity between 50-100% or more, an astonishing level of improvement. This dramatic improvement in performance, in the same range of magnitude, was also seen, for example, with the Rotarod. |
Mice were treated for 7 weeks, with clinical gerotherapeutic candidate PTC-2105 alone, with semaglutide alone, with semaglutide alone for 4 weeks, which was then stopped for the next 3 weeks, and the combination of PTC-2105 and semaglutide for 7 weeks, or in combination with PTC-2105 administered for the full 7 weeks while semaglutide was co-administered for just the first 4 weeks and then stopped for the remaining 3 weeks, or with saline negative control, before undergoing phenotyping on a number of function-testing apparatuses. Data from the Motorized Treadmill, relative to negative control saline-treated mice, which measures exercise capacity and endurance, is displayed but is representative of the findings seen across the entire phenotyping evaluation.
Not surprisingly, after only 7 weeks of treatment, PTC-2105 had just a modest positive effect on improving exercise performance, since we have learned that the elimination of senescent cells is only apparent after 8 weeks or greater of peptide treatment. Note that Group 4, treated with semaglutide for 7 weeks, experienced a significant loss of exercise endurance, presumably due to the well-known associated loss of muscle mass, and consistent with the literature. However, when co-administered, PTC-2105 worked synergistically with semaglutide, dramatically improving exercise capacity between 50-100% or more, an astonishing level of improvement. This dramatic improvement in performance at the same magnitude was also seen, for example, with the Rotarod. This led us to dig further into what could be happening.
This constellation of findings supports the hypothesis that the synergy seen between our gerotherapeutic and semaglutide is not due to elimination of senescent cells, given the short timeframe (<8 weeks) in which it was observed and its unprecedented magnitude in increased function, and must be due to an alternative mechanism. Upon further investigation, we determined that this synergistic effect between our gerotherapeutic peptides and semaglutide, seen after only <8 weeks of combination therapy (before elimination of senescent cells occurs), is due to enhancement of mitochondrial function in senescent cells, and possibly also in non-senescent cells, through targeting of both our peptides and semaglutide to the inner mitochondrial membrane. One lesser-known mechanism of action of semaglutide is that it slightly increases Mitochondrial Membrane Potential (Δψm), which, as I have already mentioned in this article, is crucial for maintaining mitochondrial function and energy production. This enhancement in mitochondrial function is also observed when our gerotherapeutic peptides are dosed as monotherapy but is less apparent due to the lower magnitude of improvement.
Mechanism #2: Increasing the Δψm in the mitochondria in senescent cells
Thus, in a second mechanism, in senescent cells, but also possibly in non-senescent cells, in combination with semaglutide but seen to a lesser degree with monotherapy, our gerotherapeutic peptides enhance the efficiency of this process in the mitochondria, increasing the proportion of potential energy in the MMP (ΔΨm) that is converted to ATP.
Unlike Mechanism #1, which takes 8 weeks or greater to observe, Mechanism #2 can be observed, for example in senescent IMR90 fibroblasts in vitro, within 24 hours, using a Seahorse Analysis to evaluate mitochondrial function.

In addition to their strong senolytic activity that capitalizes on the increased susceptibility of senescent cells to undergo apoptosis due to their lower MMP (ΔΨm), our peptides additionally gain access to the largest reservoir of untapped energy in the body, the potential energy present in efficient oxidative phosphorylation that occurs in the mitochondria in the presence of oxygen and produces 34 ATP, or “packets” of biologically useful energy, per molecule of glucose, as opposed to only 2 ATP per molecule of glucose produced during anaerobic glycolysis that occurs in the cytoplasm when oxygen is not available. This 17:1 ratio drives large gains in energy production if a cell or organism can maintain maximal mitochondrial efficiency. Unfortunately, as we age, the potential energy stored in the ΔΨm reduces with age, due to a variety of mechanisms but primarily due to “proton leak” across damaged mitochondrial membranes, reducing their efficient transformation of glucose and oxygen into ATP, the energy driving all of life’s processes. Our candidate gerotherapeutics address both the inflammaging driving aging due to senescent cells and the reduction in mitochondrial efficiency in senescent cells as we age, via a single, age-specific target, the ΔΨm.
In summary, we have discovered a number of gerotherapeutic peptides with the remarkable ability to rejuvenate naturally aged mice via two separate mechanisms, both targeting the MMP (ΔΨm) to dramatically eliminate senescent cells throughout the body, and to enhance the efficiency of mitochondrial function in senescent cells, and possibly also non senescent cells, with no apparent adverse side effects. Our peptides work via a completely novel mechanism of action that is not mediated by the binding of our peptides to any specific receptor.
What does all this mean for the field of geroscience?
We believe our comprehensive preclinical data package supports our contention that we may have discovered the world’s first safe and effective gerotherapeutics that are aimed at an aging-specific target. Why is it so important that our therapeutic target is aging-specific? The challenges of developing a gerotherapeutic designed primarily for a healthy aging population have been well covered by others. Among the many challenges, the fact that while the World Health Organization (WHO) considers aging to be a disease and has added “aging associated decline in intrinsic capacity” to the International Classification of Diseases (ICD), the FDA does not currently consider aging to be a disease that can be treated. Instead, the FDA considers aging to be a natural process, and therefore drugs that target aging must target a disease that often results from aging.
If a gerotherapeutic were to be considered for approval in healthy aging individuals and moved from the current “disease treatment” paradigm to a “disease prevention” one, which would have the greatest potential for achieving meaningful advances in healthspan, it would have to have a sterling safety track record. The problem with targeting more fundamental pathways of cell biology in an attempt to reverse the aging phenotype, is that the risks of encountering disqualifying off target side effects increase exponentially. The lower mitochondrial Δψm targeted by our gerotherapeutic peptides is highly specific to aging cells, primarily to senescent cells, and to cancer cells. Furthermore, our peptides appear to specifically target the pathologic “proton leak” that develops as we age due to accumulating damage to the mitochondrial membrane. Because of the powerful force of evolution, and because the procurement of energy to drive the processes of life that ensure survival and reproduction are so important, it is antithetical to believe that squandering any of the MMP (Δψm) through the fruitless leak of protons through the IMM without generating ATP, is of benefit to a living organism, given the effort and emphasis through so many behaviours and processes to creating the MMP (Δψm) in the life cycle of every living organism. Thus, simply reducing this proton leak, to restore the youthful integrity of the IMM, seems to be consistent with the prime directives driving life.
In our preclinical studies performed in naturally aging mice up to 90 weeks old, analogous to a 60 year old human, we have seen no indication of toxicity after over 16 weeks of therapy. In our current thinking, we believe the majority of the short and long term benefits of our gerotherapeutic peptides could be captured with just such a short course of episodic therapy, further limiting the risks of complications.
What’s next?
Our truly unique discovery has provided us with the opportunity to dramatically “jump start” the entire longevity field with an ambitious plan to bring our gerotherapeutics into human clinical trials in two stages:
Stage 1:
- Lead gerotherapeutic peptide PTC-2105 (whole body)
- Two Phase 1b/2a randomized clinical trials:
- Obesity/glucose metabolism (Clinical Indication #1) with and without GLP-1/GIP
- Sarcopenia/frailty (Clinical Indication #2)
- IND enabling studies for PTC-2107
Stage 2
- Lead gerotherapeutic peptide PTC-2107 (brain)
- One Phase 1b/2a randomized clinical trial
- Cognitive Disorders (Clinical Indication #3)
We are in the process of raising a Series A financing to fund Stage 1 of this strategic plan.
Eos SENOLYTIX slide deck registration form
Register to see Eos SENOLYTIX’s deck. By completing this form, you understand that your email address will be shared with Eos SENOLYTIX.
READ MORE: Brent Saunders appointed Advisory Board Chairman of Eos SENOLYTIX
About Kevin Slawin

Kevin Slawin, MD is the Founder and CEO of Eos SENOLYTIX, a longevity company whose lead program is focused on its proprietary gerotherapeutic peptides that have demonstrated the ability to rejuvenate naturally aged mice via two separate mechanisms (eliminating senescent cells throughout the body in the medium to longer term and enhancing the efficiency of mitochondrial function in those senescent cells in the shorter term).
He is also the founder of Perseus SENOLYTIX, an oncology company developing novel gerotherapeutic peptides to target cancer via this same novel mechanism. Interestingly, cancer cells also have less capacity to maintain the mitochondrial ΔΨm compared with normal cells, suggesting ΔΨm as a selective functional target for cancer too. In preliminary studies, the gerotherapeutic peptides demonstrated activity against TOM-1, a highly aggressive human Acute Lymphoblastic Leukemia cell line harboring a wild type ABL1 mutation. In pre-clinical studies, these peptides have been administered at escalating doses for over 16 weeks with no evidence of toxicity, opening the door for a potential safe and effective new class of cancer therapeutics.
He is also the Founder and CEO of Phoenix SENOLYTIX, a longevity company developing novel gene therapies whose lead program uses its proprietary, purposeful cell elimination technology, ApoptiCIDe™, to target senescent cells for elimination, restoring the youthful balance of aged or “senescent” and young cells throughout the organs of the body.
He was the co-founder of Bellicum Pharmaceuticals, Inc, leading it to a successful $161 million IPO in December 2014. He is also Founder, Chairman and CEO of PrintBio, Inc, the only clinical- and commercial-stage regenerative medicine company solving medical challenges with custom-engineered 3D-bioprinted living implants. Kevin currently lives in Miami, FL where he is Founder and CEO of Miami Medicos, a membership organization of physicians, founders, executives and investors catalyzing the healthcare entrepreneurial ecosystem in Miami and worldwide.


